Answer Question Screen
- Clean multiple-choice interface with progress bar.
- Mark for review feature.
- Matches real test pacing.

** All Prices are in US Dollars (USD) **
Understanding the exact breakdown of the NYSTCE CST Chemistry test will help you know what to expect and how to most effectively prepare. The NYSTCE CST Chemistry has 90 multiple-choice questions and 1 essay questions. The exam will be broken down into the sections below:
| NYSTCE CST Chemistry Exam Blueprint | ||
|---|---|---|
| Domain Name | % | Number of Questions |
| Structure of Matter | 20% | 18 |
| Properties of Matter | 20% | 18 |
| Chemical Reactions | 20% | 18 |
| Energy | 20% | 18 |
| Pedagogical Content Knowledge (Constructed-Response) not on Practice test | 20% | 18 |
Everything you need to prepare with confidence—without wasting a minute.
Timed, No Time Limit, or Explanation mode.
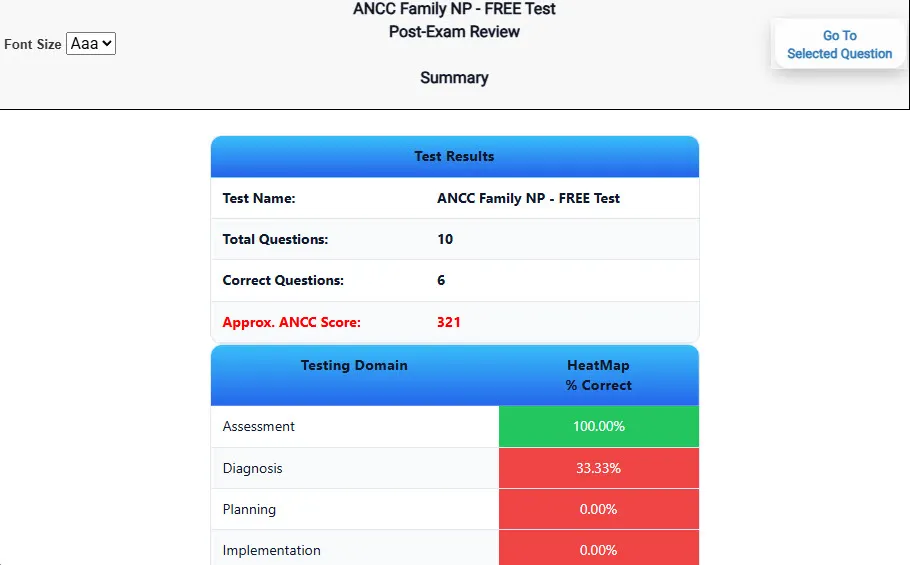
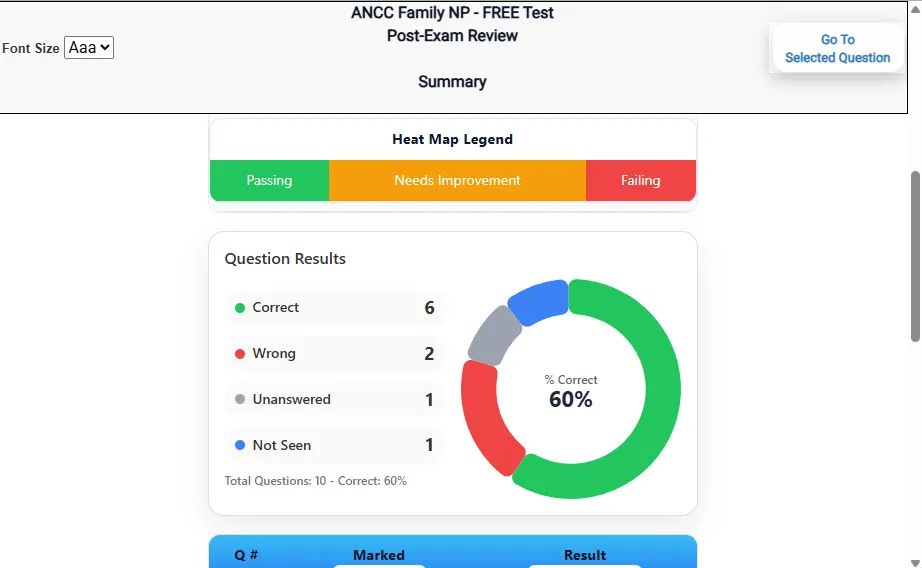
Heatmaps and scaled scores highlight weak areas.
Concise explanations emphasize key concepts.
Matches the feel of the actual exam environment.
Clean layout reduces cognitive load.
Web-based access 24/7 on any device.

Our practice tests are built specifically for the NYSTCE CST Chemistry exam — every question mirrors the real topics, format, and difficulty so you're studying exactly what matters.
We match the per-question time limits and pressure of the actual NYSTCE exam, so test day feels familiar and stress-free.
You'll have more than enough material to master every NYSTCE CST Chemistry concept — no repeats, no fluff.
Ordering 5 practice exams costs less than retaking the NYSTCE CST Chemistry exam after a failure. One low fee could save you both time and money.
Need to step away mid-exam? Pick up right where you left off — with your remaining time intact.
See your raw score and an estimated NYSTCE CST Chemistry score immediately after finishing each practice test.
Review correct and incorrect answers with clear, step-by-step explanations so you truly understand each topic.
We're fully accredited by the Better Business Bureau and uphold the highest standards of trust and transparency.
No software to install. Access your NYSTCE CST Chemistry practice exams 24/7 from any computer or mobile device.
Need extra help? Our specialized tutors are highly qualified and ready to support your NYSTCE exam prep.
Preparing for your upcoming NYSTCE CST Chemistry (161) Certification Exam can feel overwhelming — but the right practice makes all the difference. Exam Edge gives you the tools, structure, and confidence to pass on your first try. Our online practice exams are built to match the real NYSTCE CST Chemistry exam in content, format, and difficulty.
These NYSTCE CST Chemistry practice exams are designed to simulate the real testing experience by matching question types, timing, and difficulty level. This approach helps you get comfortable not just with the exam content, but also with the testing environment, so you walk into your exam day focused and confident.
“ Not a user friendly experience
“ Didn't really reflect the questions that were on the test for the most part. Also, they totally got the format of the constructed response question wrong, which never changes. Overall OK if you have nothing else, but definitely not worth the price they charge in my opinion.
“ It was a good test run. Now I know what to expect and what I need to study.
“ The experience was very practical and I did well.
“ Very easy to navigate.
“ Exam Edge was terrific. Even better than I hoped. Just completing and reviewing the tests are a great learning experience. I can't wait to take the certification test. Tom S.
Here is a list of alternative names used for this exam.